What is Electrolysis
Electrolysis is a technique that uses applied electric current to drive non-spontaneous chemical reactions. Electrolysis allows the interchange of atoms/ions by the removal or addition of electrons. Electrolysis involves two electrodes (an anode and a cathode). When direct current (DC) is applied, oxidation occurs at the anode and reduction occurs at the cathode.

In membrane salt splitting electrolysis, the electrodes are submerged in electrolyte solution(s) that are hydraulically separated by one or more ion exchange membranes. When direct current (DC) is applied, the following reactions occur at the electrodes:
At the cathode:
2 H2O + 2e- → 2 OH- + H2
At the anode:
H2O → 2H+ + 1/2 O2 + 2e-
The current flows through the cell by the flow of ions across the ion exchange membrane(s). The ions making up the salt are therefore separated and converted into their corresponding acid and base forms through the electrochemical process.
Lithium chloride electrolysis is a different form of membrane electrolysis. In the case of lithium chloride electrolysis, the following reactions occur at the electrodes:
At the cathode:
2 H2O + 2e- → 2 OH- + H2
At the anode:
2Cl- → Cl2 + 2e-
Though there is water present at the anode.
Whether oxygen or chlorine is generated at the anode depends on the anode compartment electrolyte (referred to as the anolyte) and the anode material.
The system consisting of the anode, cathode and ion exchange membrane(s) is referred to as an electrochemical cell. A stack of connected cells is coined "electrolyser".
Electrolysis in Action
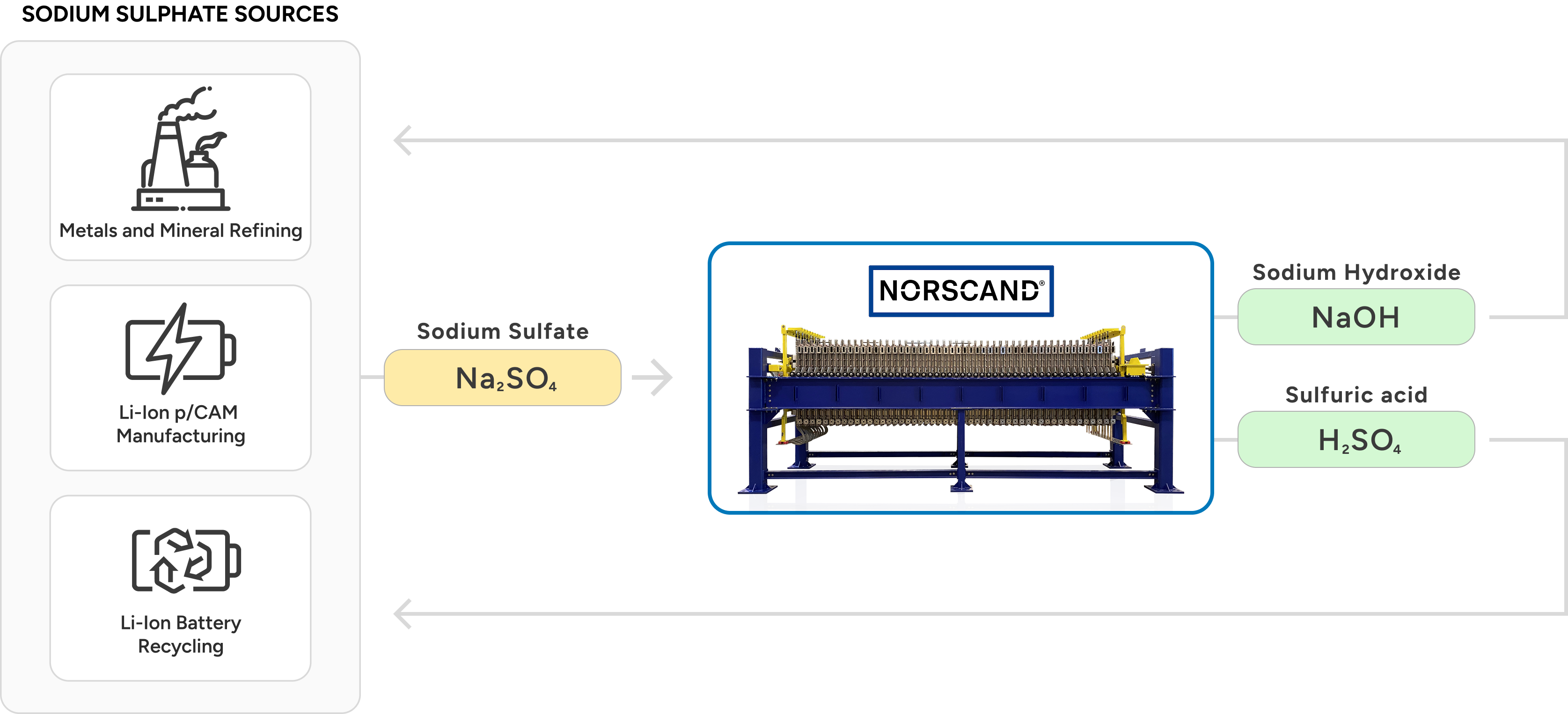
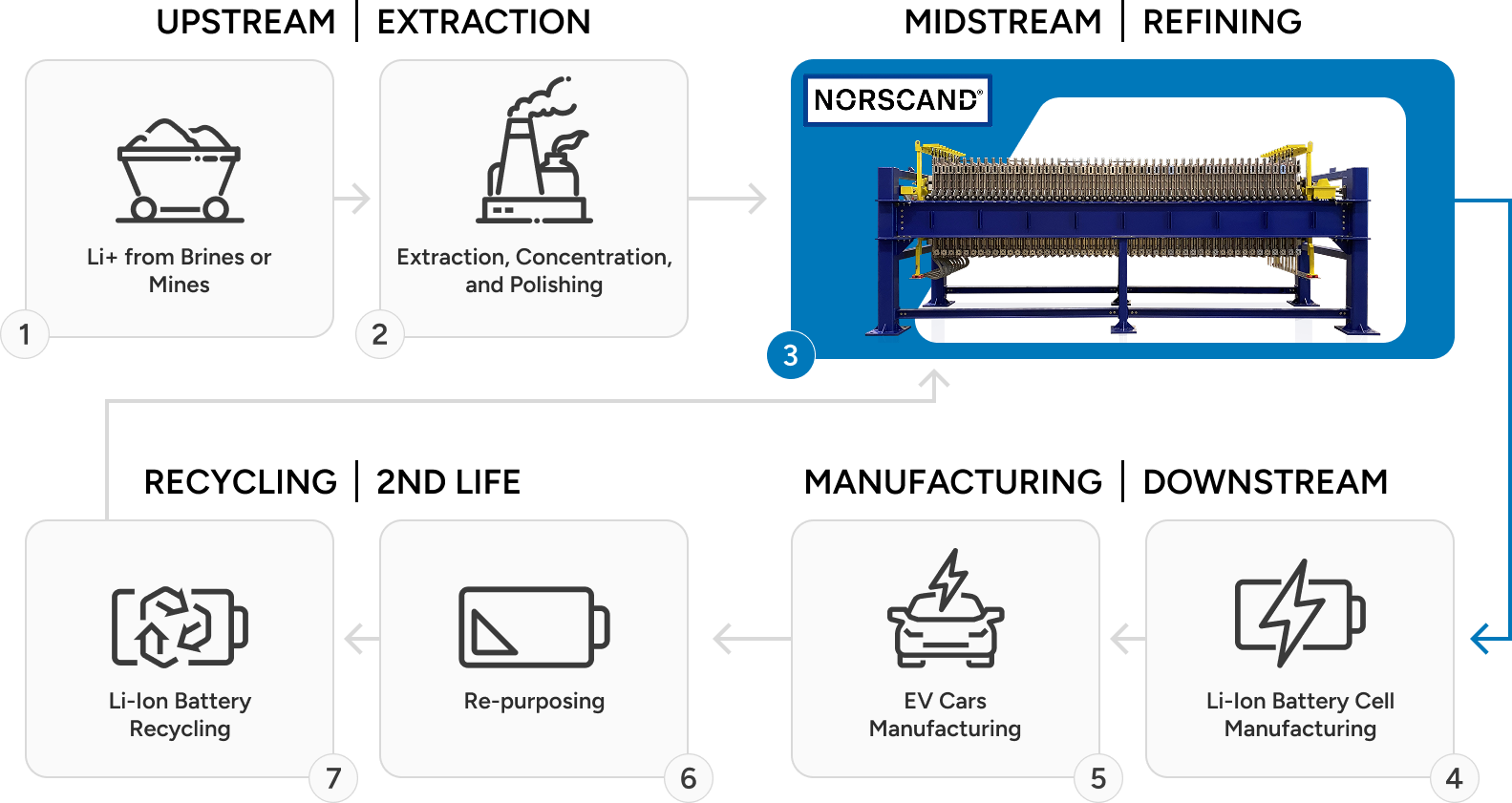
From sodium sulfate recovery to lithium battery lifecycle management, NESI's electrolysis platform enables sustainable, closed-loop chemical conversion processes across upstream and downstream industries.

Key Features
-
Electrolytes: The typical cathode compartment electrolyte (referred to as the catholyte) is a monovalent cation hydroxide such as lithium hydroxide or sodium hydroxide. The range of anolyte solutions is more diverse as it is dependent on the brine feed and the number of hydraulic compartments in the cell. Typical anolyte solutions include lithium chloride, sodium bisulphate, lithium bisulphate, and sulphuric acid.
-
Ion Exchange Membranes: The hydraulic compartments within the cell are separated by one or more ion exchange membrane(s). An ion exchange membrane is selectively permeable meaning that it only lets specific charged ions pass through depending on charge of the ion. Typical two compartment salt splitting electrolysis will have a single cation exchange membrane separating the anode and cathode compartments. Typical 3 compartment salt splitting electrolysis will have a cation exchange membrane and an anion exchange membrane separating the anode, centre/brine, and cathode compartments.
-
Temperature and Pressure: Standard operating temperatures of 50 to 80 °C, near atmospheric pressures.
-
Current Density: 2000 to 5000 A/m2 (higher than similar technologies like bipolar membrane electrodialysis.
Applications
- Production of lithium ion battery precursor materials (i.e. lithium hydroxide monohydrate) from lithium ion battery recycling, salars or hard rock processing.
- Regeneration of valuable feedstocks from waste salt solutions (e.g. production of sulphuric acid and sodium hydroxide from sodium sulphate waste).
Discuss Your Application
Our experts can help you find the right electrochemical solution for your process.
Contact Us